iWand Manual Radionuclide Applicator System

Manual Radionuclide Applicator System
Liberty Vision Corporation has designed and manufactured an FDA 510(k) cleared high-dose-rate beta radiation disc source (the LV Y‑90 Disc). To facilitate disc placement on the eye, two FDA Class-I hand-held devices are available: iWand® A (for Anterior placement) and iWand® P (for Posterior placement).
Each was specifically designed to safely optimize episcleral application of the LV Y‑90 Disc.
KEY FEATURES:
Extraocular, minimally invasive
Hand-held applicators facilitate radiation placement
Accurate, reproducible source positioning
Minimal side scatter to healthy tissues
A single-use quick treatment

iWand® A — For Anterior Use
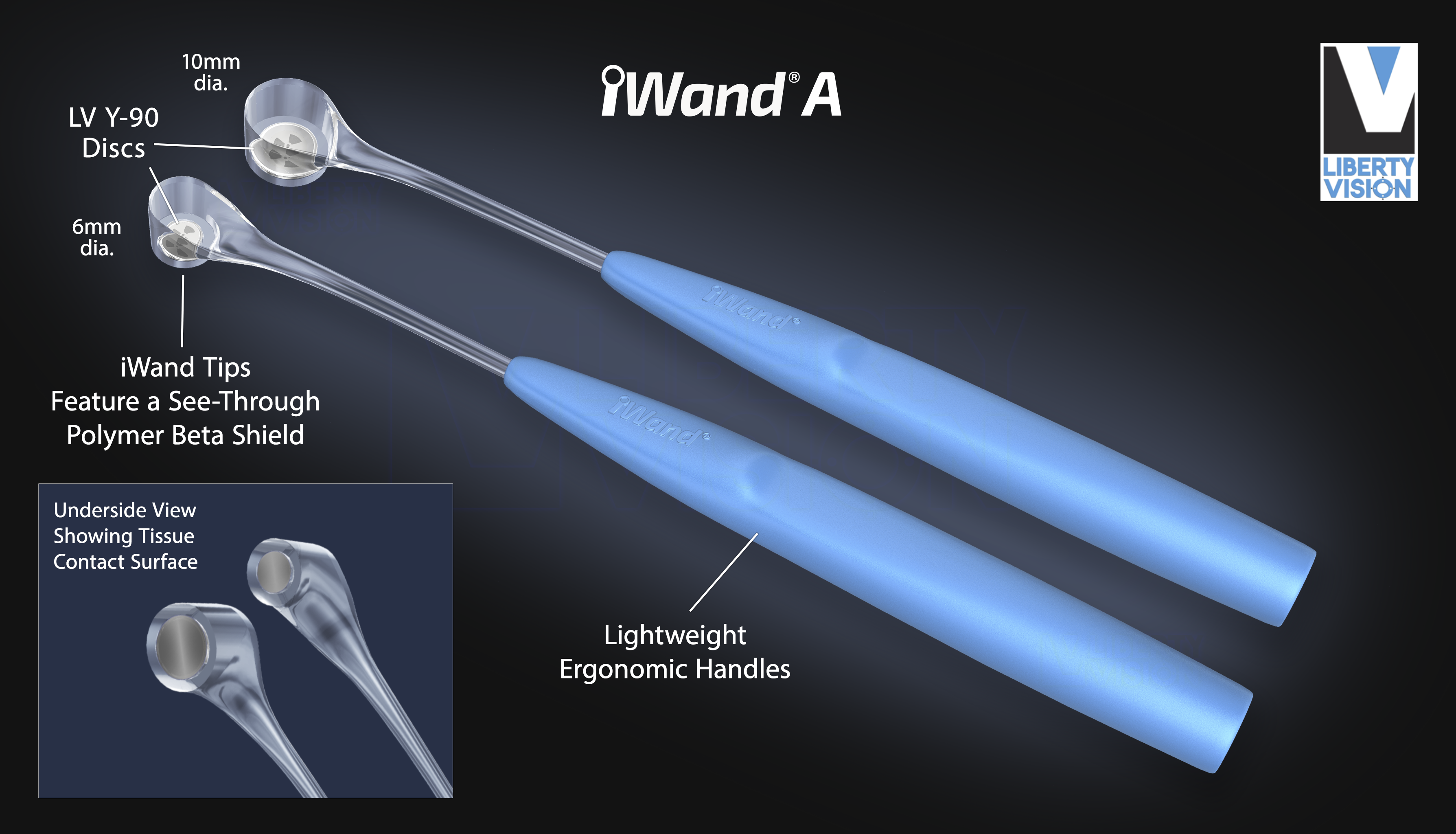
The iWand A was designed for episcleral treatment of anterior segment ocular diseases with beta radiation using the LV Y‑90 Disc. The lightweight, ergonomic iWand A radionuclide applicator can be directly applied to the anterior ocular target, with or without prior surgery. The anterior segment applicator features an advanced see-through polymer that aids in shielding the surgeon from radiation exposure, while allowing a view of the ocular target during treatment. Each iWand A hand-held applicator is pre-sterilized and individually packaged for single use. This unique Liberty Vision system offers unique and helpful features. The LV Y‑90 Disc's short half-life is environmentally friendly and facilitates disposal.
Learn more about the iWand A
iWand® P — For Posterior Use

The iWand P was designed for episcleral treatment of posterior ocular tumors and benign growths. Optimized for extraocular placement of beta radiation, the iWand P holds the patented LV Y‑90 Disc. Therefore, it requires minimal surgery for exact placement. Liberty Vision makes viewing of the disc behind the eye possible by incorporating four surrounding guiding lights around the LV Y‑90 Disc. Utilizing ophthalmoscopy (indirect or photographic) to view those four lights during implantation, the surgeon positions and localizes the disc over the targeted zone. The iWand P lights circumscribe the target, and offer direct, recordable confirmation of LV Y‑90 Disc's location during treatment. Each superlight and ergonomic iWand P is individually packaged and sterile for single use.
Learn more about the iWand P