
Pilot Production Begins
Pilot production begins at Liberty Vision.

Pilot production begins at Liberty Vision.

Liberty Vision CEO Dr. Paul T. Finger contributes to a new book on surgical ophthalmic oncology.
Liberty Vision secures larger facilities to support growing operations.

Testing and qualification of device production and processes is underway.

Several key US patents granted for the iWand® device, its key technologies, and variants.

Patents granted for the Liberty Vision iWand® source in the US and Europe.

Liberty Vision manufacturing start-up begins.
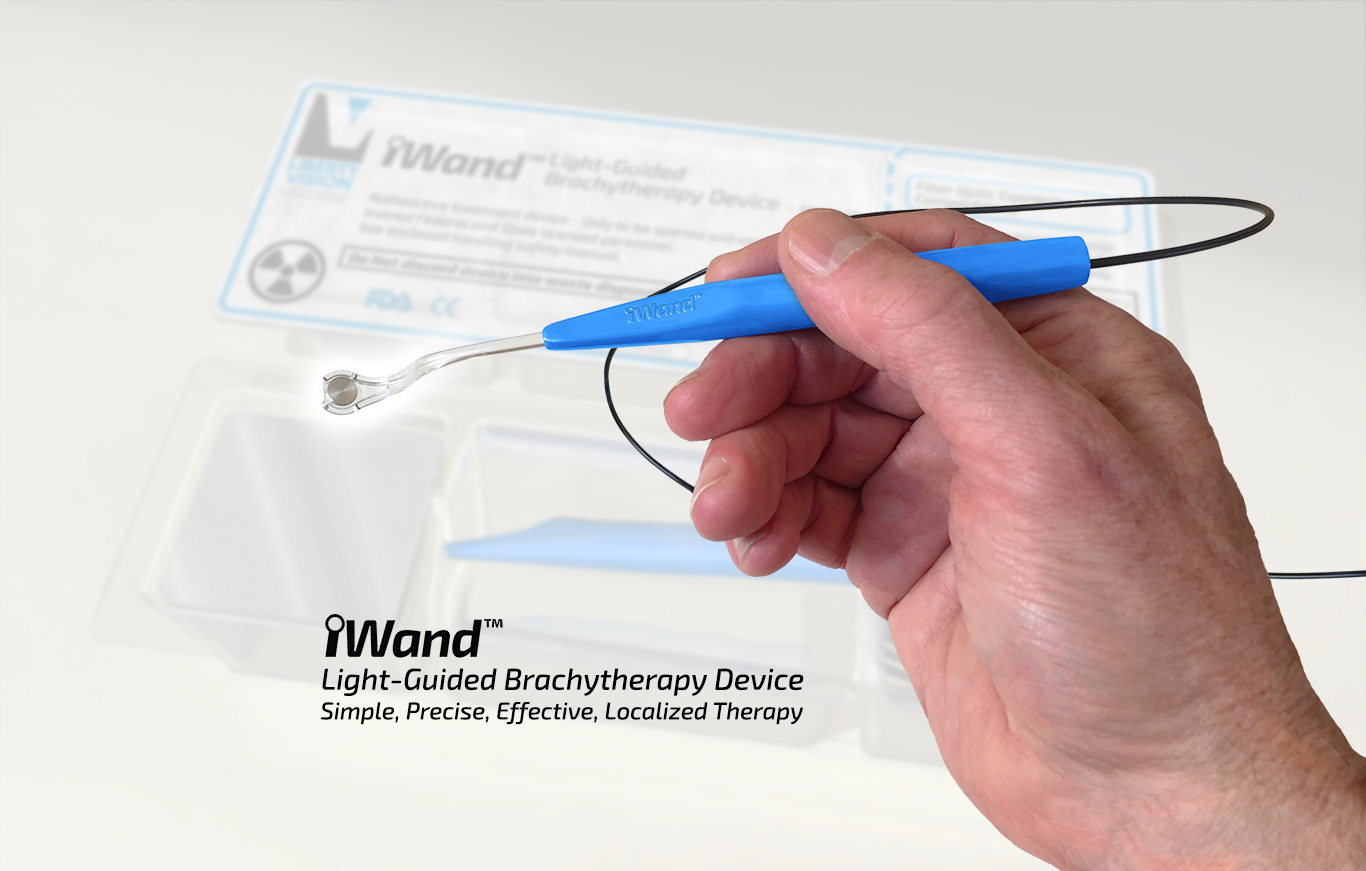
IP refinement completed and multiple additional patent applications submitted.
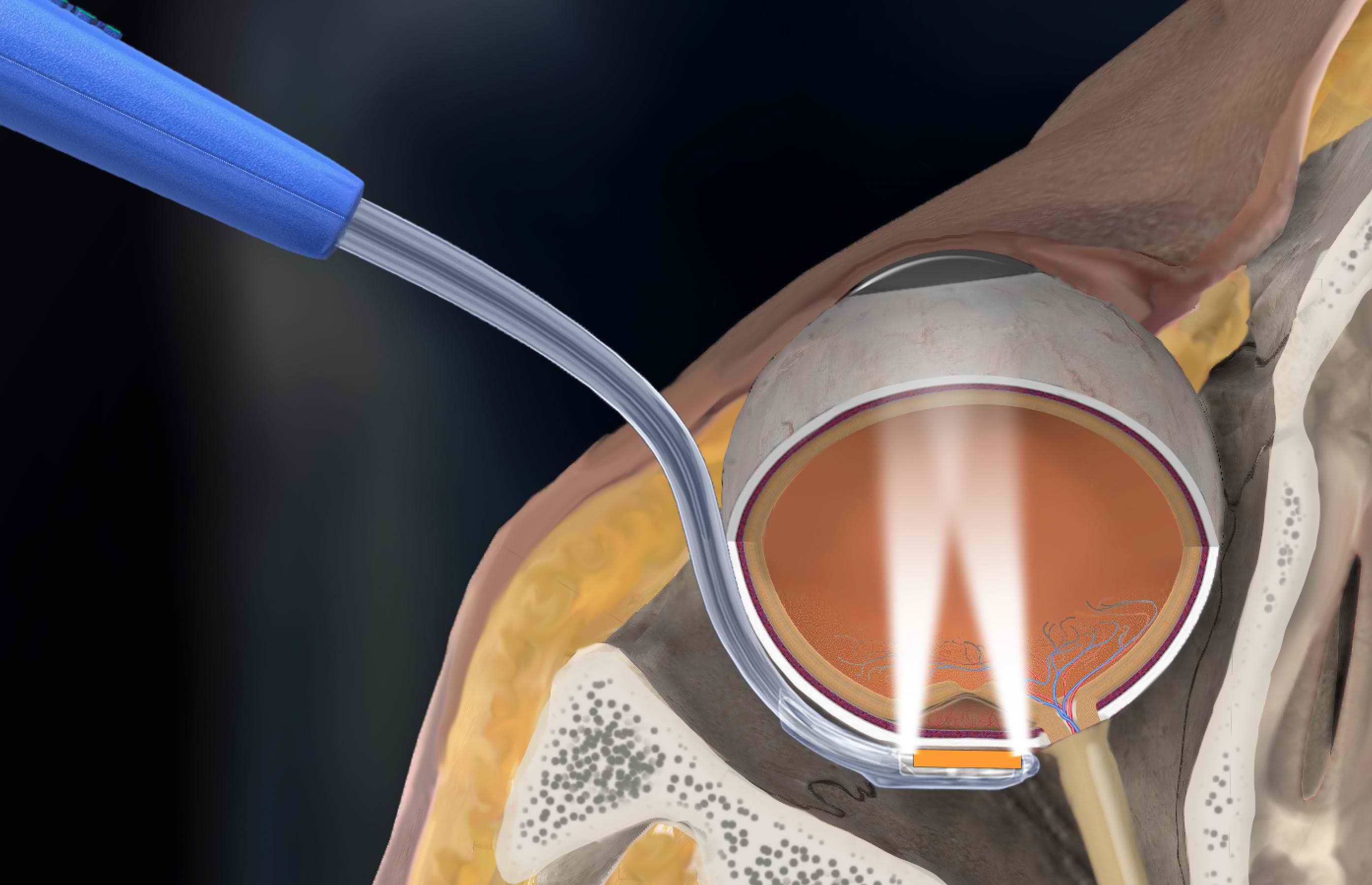
Liberty Vision completes physics testing of the iWand device.
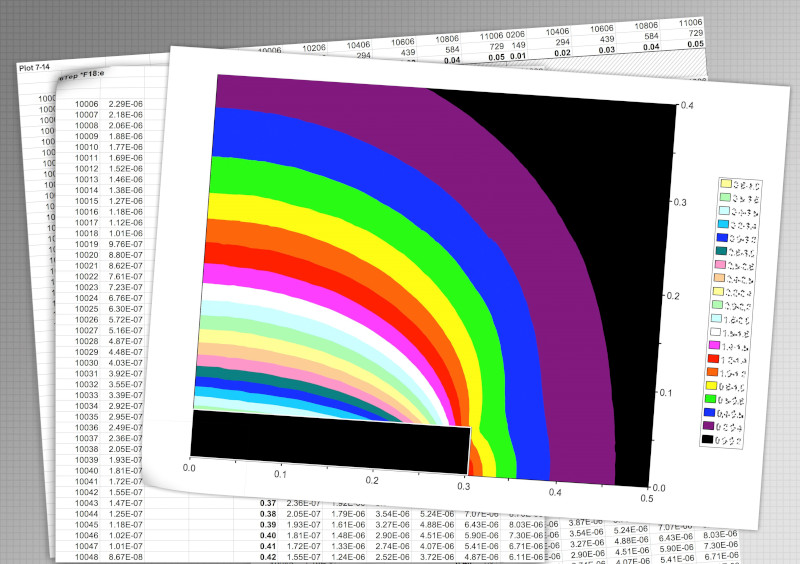
Liberty Vision ramps up the radiological R&D phase following a successful product design phase.